Trial Design Stage
Published Date: April 29, 2025
Share to:
Helpful Terms and Definitions
*This list is not exhaustive, nor does CTTI officially endorse the resources created by other organizations.
Draft Protocol
Questions To Ask
- Why is this study appealing to the patient community?
- Is the flow burdensome for patients?
- Are the inclusion/exclusion criteria accurate to the current patient population?
- Are these the endpoints and outcomes that matter most to the patients?
- How is patient health information being protected and used?
- What does the risk to benefit analysis look like?
- What types of control groups and treatments are being used?
- What are the options for patients whose disease worsens during treatment?
- What is the long-term strategy - is there an open label extension period?
- How and when are patients diagnosed?
- What type of providers are giving care?
- What are barriers to accessing treatment? Are there solutions?
Why involve tech, device, and data companies now?
During the draft protocol stage, collaboration with technology, device, and data partners helps ensure that study design elements can be operationalized in a feasible, scalable, and data-driven manner.
Their involvement supports the use of real-world data and analytics to inform patient population feasibility, refine study criteria, and anticipate recruitment and retention challenges. Early engagement also helps reduce the risk of protocol amendments by aligning data capabilities, study requirements, and operational workflows before finalization.
What aspects of the draft protocol should they inform?
- Patient population feasibility: Use real-world data sources to help characterize potential study populations and assess recruitment feasibility
- Diversity and representation planning: Inform targets using epidemiologic and real-world data insights
- Inclusion and exclusion criteria optimization: Assess the impact of eligibility criteria on population size, diversity, and feasibility
- Recruitment feasibility and site strategy: Identify geographies and sites with access to eligible patient populations
- Predictive feasibility modeling: Inform projections such as enrollment timelines, sample size assumptions, and dropout risk
- Retrospective and real-world data analysis: Identify potential risks (e.g., adherence challenges, safety considerations) based on historical or real-world evidence
Potential When and How to Engage with These Technologies
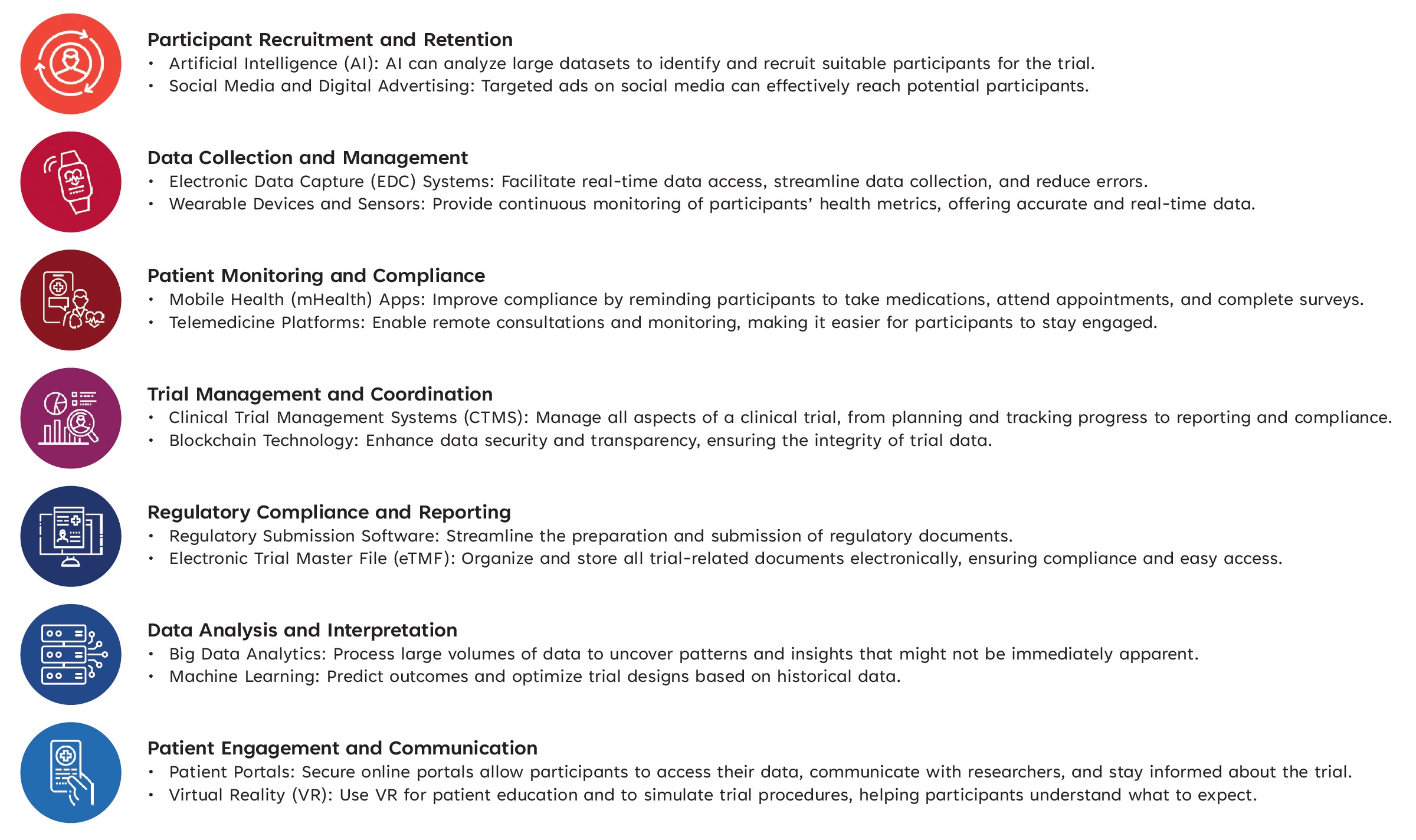
How technology may support execution (high-level examples)
- Data capture and management (e.g., EDC, remote data collection tools)
- Patient engagement and monitoring (e.g., apps, telehealth, wearables where appropriate)
- Trial operations and oversight (e.g., CTMS, centralized monitoring)
- Data analysis and modeling (e.g., analytics platforms supporting feasibility and outcomes analysis)
Tech, Device, and Data Company Resources
A Smarter Approach to Clinical Trial Design and Delivery
Using digital technologies in clinical trials: current and future applications
Embracing Hybrid Trial Design - How, When, & Why To Use It
Artificial Intelligence for Clinical Trial Design
Clinical Trial Management Systems
 Patient and Caregiver
Patient and Caregiver Investigator
Investigator Regulator
Regulator IRB
IRB Tech, Device,
Tech, Device, Payer
Payer