Topics Included: Data Collecting and Reporting, Innovative Trials, Regulatory Submissions + Approvals
SHARE TO:
Overview
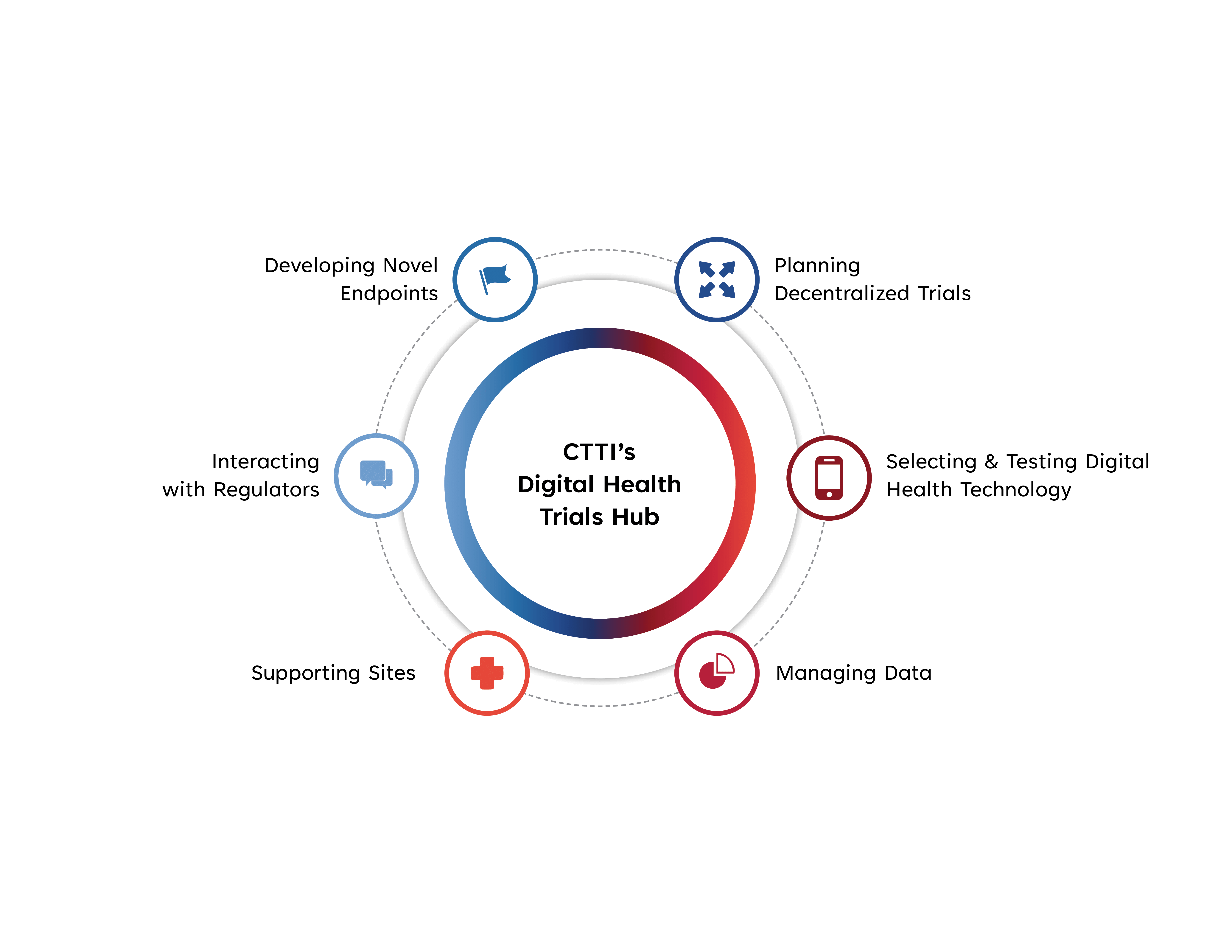
Digital health trials are here to stay. Use CTTI's broad suite of recommendations and resources to design and run a successful, fit-for-purpose digital health trial that meets your research goals.
Areas of Focus
DEVELOPING NOVEL ENDPOINTS
Develop novel endpoints that more accurately represent the patient experience – and therefore – may be more meaningful to patients, providers, and others.
PLANNING DECENTRALIZED TRIALS
Learn about the foundational recommendations for designing and running a fit-for-purpose decentralized clinical trial.
SELECTING & TESTING A DIGITAL HEALTH TECHNOLOGY
Understand what needs to be considered and measured before selecting a digital health technology.
MANAGING DATA
Understand, plan for, and address the new challenges associated with managing data from digital health technologies.
SUPPORTING SITES
Develop a robust digital health technology management plan, accounting for testing, documentation, technical support, training, communication, data integrity, and participant safety.
INTERACTING WITH REGULATORS
Develop an appropriate strategy for collecting and sharing digital health trial data with regulatory bodies.
This Hub is a collection of recommendations and resources that have been created by multi-stakeholder teams. In the development of the Hub, CTTI has repackaged four original Digital Health Trials (formerly known as Mobile Clinical Trials) projects. Historical information including project team members and deliverables from each of these projects can be found here.
