Introduction
The following recommendations aim to highlight the unique benefits of using disease progression modeling in medical product development, when it should be considered over other tools or approaches, and provide considerations for what is needed and how to incorporate it.
These recommendations are intended for decision makers in medical product development, including chief medical officers, heads of innovation or translational medicine, leadership roles in therapeutic areas, project team leads, and regulatory affairs.
Cross functional leaders often encounter similar questions around internal resources, evidence prioritization, and return of investment when making medical product development decisions. These recommendations will help decision makers ask the right questions of modeling experts and other subject matter experts to recognize the value of DPM and inform decisions about its implementation.
What is disease progression modeling?
A disease progression model (DPM) is a mathematical model that quantitatively describes the time course or trajectory of a disease.
Disease progression modeling can link disease progress, treatment effect, and/or patient behavior to clinical trial outcomes and inform decision making throughout the medical product development process.
While some modeling and simulation approaches such as pharmacokinetic/pharmacodynamic (PK/PD) and physiologically based pharmacokinetic (PBPK) modeling are well recognized as enabling clinical trials and model-informed drug development, the potential of disease progression modeling to improve the quality and efficiency of medical product development has yet to be fully realized.
How can disease progression modeling impact medical product development?
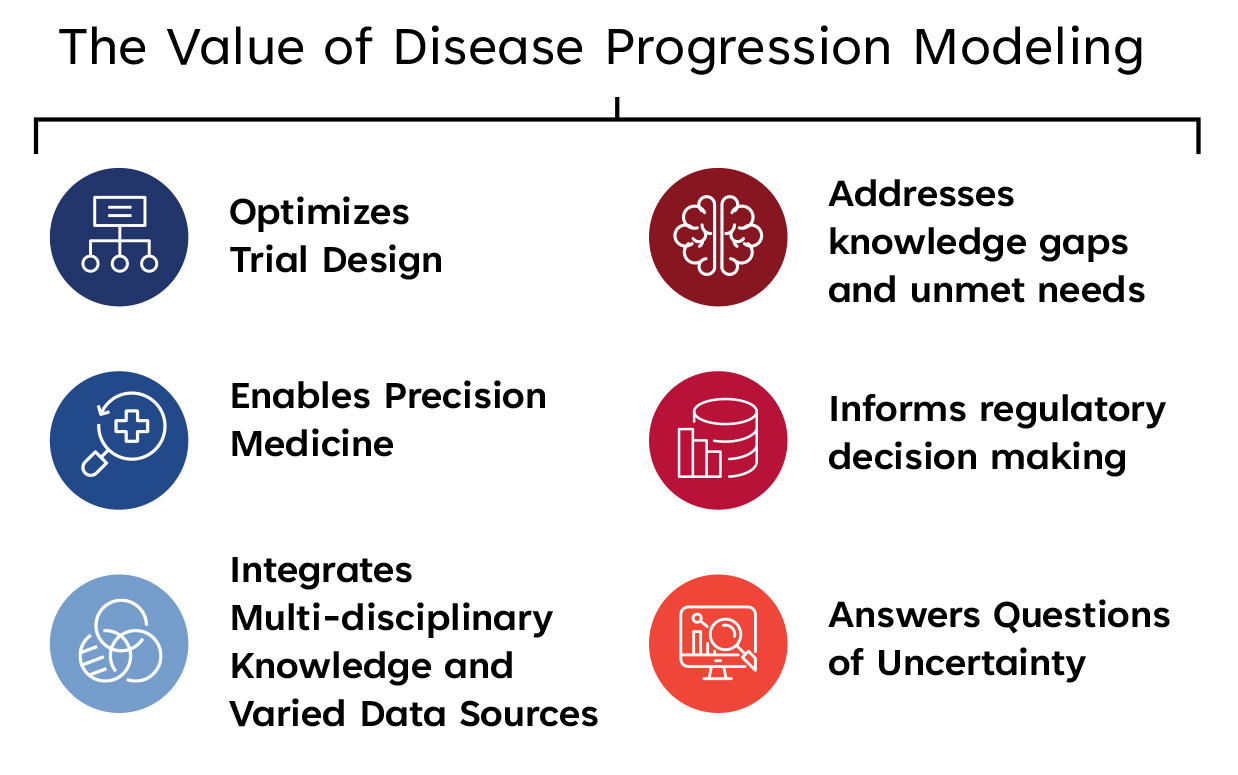
The use of a DPM integrates multi-disciplinary knowledge and data from different sources, including translational, clinical trial, and real world data to:
- Impact trial efficiency, especially for trials with diseases that progress over a long duration of time, for which running a clinical trial during that period of time is costly and burdensome.
- Answer questions of uncertainty. It can account for and provide clarity around heterogeneity across patients and at an individual level.
- Tailor trials towards precision medicine. Recognizing that diseases progress differently, across populations and in different stages within a patient, DPM can address individual factors or covariates affecting progression to enable trial design optimization and tailor trials towards precision medicine (e.g. stratify populations or adapt treatment plans).
- Address unmet needs and knowledge gaps, including supplementing for a small population size with digital twins in rare disease trials, or stratifying a population for patient enrichment for diseases with heterogeneous phenotypes (e.g. autoimmune and neurodegenerative diseases), or supporting a limited understanding of the disease by modeling disease course for endpoint selection.
- Inform regulatory decision making. DPM contributes to the totality of understanding of a medical product’s benefits and risks, helping to advance its development and support regulatory decision-making both in premarket product review and post-market product assessment.
CTTI further describes the value of DPM, its current applications, and opportunities for uptake in the following article published in Clinical Pharmacology & Therapeutics called The Potential of Disease Progression Modeling to Advance Clinical Development and Decision Making.
Recommendations Summary
- Evaluate if longitudinal data about the disease course is critical to answer a question of interest for medical product development decisions.
- Assess the value of using a disease progression model compared with other potential approaches.
- Use the most up-to-date understanding of the disease and measure(s) of progression available to inform your disease progression modeling approach.
- Determine whether the necessary sources of data are available, relevant and reliable to develop a disease progression model.
- Determine whether technological and model resources, as well as the right skill sets, exist or can be obtained to support the disease progression model development and implementation.
- Begin using disease progression modeling early on in the medical product development lifecycle to build confidence in the model for later stages of development.
- Continually assess the performance and ensure the disease progression model is qualified for the intended application.
- Leverage new technology and evolving methods to advance uses of disease progression models.
- Disseminate critical insights on disease progression modeling to promote standardized practices and foster confidence.
Recommendations








