QbD Toolkit: Overview, Learn About QbD , Teach Others About QbD, QbD Principles Document, Exploring the CTQ Factors, Workshop Tools, Adopt QbD, How Others are Using QbD, Get Started
QbD Toolkit Resources: Principles Document, CTTI QbD Recommendations, Key Stakeholders, Maturity Model, Metrics Framework, Case Studies
SHARE TO:
This section of the Toolkit provides an introduction to QbD through videos, downloadable presentations, and peer-reviewed articles. Learn about QbD and why it matters in clinical trials. Leverage these tools to teach others in your organization about QbD in order to secure their interest and support.

CTTI's QbD Recommendations
The CTTI QbD project has produced recommendations on the use and implementation of QbD.

PowerPoint describing QbD
This PowerPoint Slide presentation provides an overview of QbD. It can be downloaded and used to teach your team about QbD.
QbD Publication in DIJ
This publication, Clinical Trials: Rethinking How We Ensure Quality, by Landray, et al. in Drug Information Journal 46(6) 657-660, provides an overview of QbD in Clinical Trials.

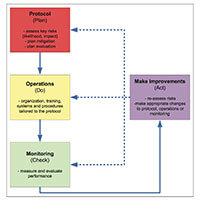
 QbD is about prospectively examining the objectives of a clinical trial and defining those factors that are critical to meeting those objectives. This requires thinking differently about clinical trials. In order to do that effectively, we have provided tools below to help introduce your team to QbD concepts and how they apply in clinical trials. The sections include:
QbD is about prospectively examining the objectives of a clinical trial and defining those factors that are critical to meeting those objectives. This requires thinking differently about clinical trials. In order to do that effectively, we have provided tools below to help introduce your team to QbD concepts and how they apply in clinical trials. The sections include: 