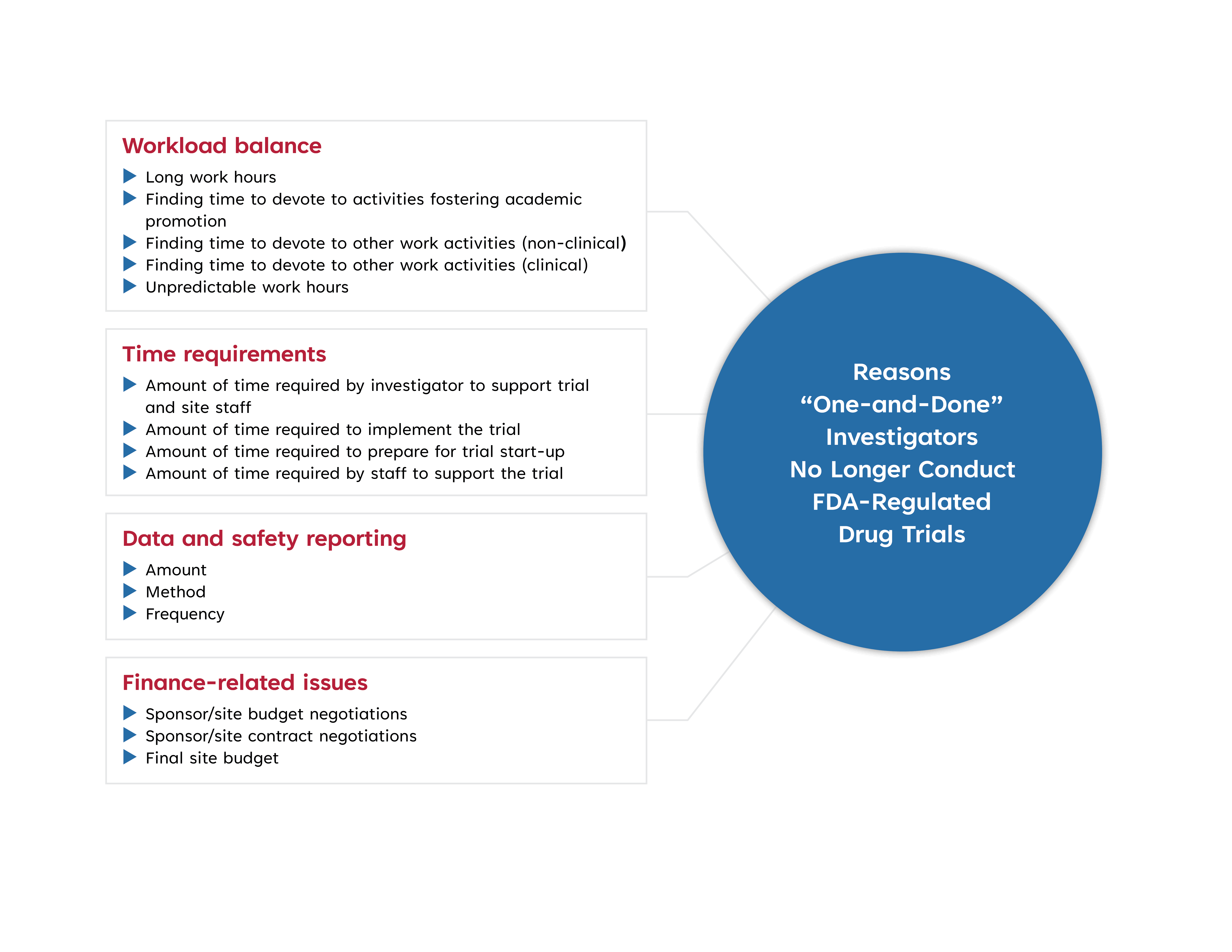
Results from a CTTI survey of “one-and-done” investigators who ended their involvement as a site principal investigator in research after a single clinical trial revealed four main reasons: 1) excessive time commitments required, 2) burden of data and safety reporting, 3) trial responsibilities taking away from other activities, and 4) dissatisfaction with study finances. Site investigator interviews were also conducted to identify other reasons why investigators have chosen to continue conducting FDA-regulated drug trials.
These results were presented and discussed at a multi-stakeholder expert meeting held on Apr. 5, 2017, and published as a peer-reviewed research article. The consensus findings from this meeting were used to develop a set of recommendations aimed at reducing attrition rates for U.S. clinical trial investigators and strengthening the field of qualified and productive investigators.
An additional investigation is underway, examining information contained in the FDA’s BMIS database, which houses electronic data from all Form FDA 1572s submitted by investigators prior to participating in FDA-regulated drug trials. These analyses are focused on characterizing temporal trends in investigator participation in FDA-regulated clinical trials, and developing a “phenotype” of characteristics that distinguish “one-and-done” investigators from those who participate intermittently or continuously in clinical research. Read the manuscript reporting on results from these analyses.