
State of Clinical Trials: Perspectives, Problems, and Pathways Forward
In May, CTTI convened 150 leaders from across the clinical trials enterprise in Washington, D.C., for a landmark State of Clinical Trials meeting. This gathering focused on the current landscape of clinical research across the five pillars of CTTI’s Transforming Trials 2030 vision, which envisions a future where clinical trials are patient-centered and easily accessible, fully integrated into health processes, designed with a quality approach, maximally leveraging all available data, and improving population health.
Key Insights and Innovations
In response, attendees identified bold solutions: empowering federated learning networks, enabling AI-driven data sharing, and creating policies that expand access and control of data. They called for making incentives more transparent, integrating trials thoughtfully into routine practice, and using storytelling to humanize trial communication. Participants also stressed the importance of engaging payers, medical students, and emerging professionals to build a more accessible and sustainable research enterprise. Click here to read the in-depth report.
Accelerating Impact: Persistent Challenges and Solutions
CTTI remains committed to leading efforts that modernize clinical trials, transforming them into efficient, transparent, participant-centered, and data-driven endeavors. Breakout sessions surfaced four persistent challenges and solution sets, including:
- Underutilized sites: Move beyond project-by-project funding to long-term investment in research infrastructure, or de-risk selection of new researchers by educating community centers collectively and focusing on fitness for purpose by role.
- Trial inefficiency and complexity: Clarify roles and train clinicians and staff to navigate dual roles transparently and ethically or recognize trial heterogeneity and categorize trials so metrics of quality and efficiency are meaningful.
- Unused data: Develop federated data platforms and centralized frameworks with participant consent integrated into the process.
- Eroding public trust: Institute patient needs assessments and humanize trial communication or enhance transparency and shared goals through consensus on what truly confers competitive advantage.
These insights reflect the expertise of our collaborative network and the vital role of our members in driving progress across all five pillars of CTTI's Transforming Trials 2030 vision. As we look ahead to 2026 and beyond, we are energized to create even more opportunities with members to lead, innovate, and shape the future of clinical trials.
AI Public Workshop - The Conversation Evolves
This year, the FDA and CTTI cohosted the second hybrid public workshop on the use of AI in drug and biological product development, bringing together regulators, industry leaders, academic researchers, and patient leaders to help move the field from promise to practice. Speakers underscored the urgency of improving outcomes while protecting patients, aligning on a risk-based credibility framework, instituting stronger data governance, creating transparency across the model lifecycle, and facilitating earlier, faster FDA–sponsor engagement so AI can be used responsibly to help streamline decisions from protocol design to pharmacovigilance.
Participants converged on a few non-negotiables: high-quality, representative, and accessible data; user-centric design with humans in the loop; globally harmonized terminology; and clear documentation to earn trust. Patient leaders emphasized transparency, usability, and inclusion so AI streamlines access and fairness rather than widening gaps. Some made calls to publish negative results.
Additionally, several specific solutions emerged in key areas:
- Access to data: create models and federated learning to share signals without sharing raw data, standing up precompetitive consortia to build and openly evaluate models, and operationalizing standardized, privacy-preserving data pipelines.
- Biomarkers: expand patient-governed “sandboxes” to co-design endpoints that matter and increase use of digital biomarkers to detect disease earlier.
- Workflow: deploy human-in-the-loop AI for high-burden workflows (site monitoring, case deduplication, safety signal triage) with auditable reasoning; consider CRA agentic copilots to cut manual reconciliation; use digital twins for robust covariate adjustment.
- Regulator engagement: Align work to the FDA’s risk-based framework. Panelists urged routine early check-ins with the FDA, implementing shared playbooks and benchmarks, and clear impact metrics (e.g., cycle time, quality, inclusion) to help advance clinical development.

CTTI Salon Dinners: Sparking Insightful Dialogue Across the Clinical Trials Enterprise
In 2025, CTTI continued its tradition of hosting dinners designed to foster open dialogue and fresh thinking among clinical trials leaders. Held alongside major industry conferences, these evening events provide a unique space to explore emerging topics and pre-competitive opportunities.
Integrating Trials into Routine Care
During CTTI's Salon Dinner at the SCOPE Summit in February, Joe O’Rourke, Head of Commercial Development and Real-World Evidence at Walgreens, shared insights on Walgreens’ evolving role in clinical research. The discussion focused on community-centered approaches, sustainable infrastructure, and the importance of partnerships, highlighting barriers to trial participation and strategies for scalable biospecimen collection and long-term follow-up.
AI/ML in Drug Development
In June, CTTI held a Salon Dinner at the DIA Global Annual Meeting, featuring Dr. Tala Fakhouri, then Associate Director for Data Science and Artificial Intelligence at the FDA’s Center for Drug Evaluation and Research. Dr. Fakhouri led a discussion on the FDA’s draft guidance for AI/ML in drug development and future directions in this rapidly evolving field, sparking conversation around data sharing, participant expectations, and balancing innovation with trust.



CTTI Fall Member Meeting: Balance in the Regulatory Ecosystem
In Fall 2025, CTTI convened members to discuss the evolving regulatory and operational landscape, focusing on regulatory balance and modernization. The FDA’s Mike Davis, CDER Deputy Director, emphasized transparency, collaboration, and openness to questioning established practices to improve efficiency. Discussions also highlighted that sponsors and other stakeholders sometimes hesitate to use available regulatory flexibilities, often due to uncertainty about acceptance or operational challenges.
Selective Safety Data Reporting: Members explored the distinction between “need to know” and “want to know” in safety data collection. For interventions with well-established safety profiles, exclusion of certain traditional safety endpoints can be a rational, patient-centered approach when justified and agreed upon by all stakeholders. Panelists emphasized that monitoring and responding to known safety issues remains essential, but this does not always require comprehensive collection and reporting of all safety data.
Trial Design Flexibility: The meeting highlighted the value of sharing and replicating flexible trial models. Strategies discussed included investing in mobile units, encouraging innovation, addressing operational barriers (such as form 1572), and building infrastructure in communities. Ensuring data integrity and maintaining collaborative dialogue among sponsors, regulators, patient groups, and sites were identified as essential for responsibly implementing new approaches. Ultimately, the research enterprise must find ways to offer trials where people are.
RWE and Causal Inference: The final session underscored that the value of data lies in its use. Members discussed robust statistical design and the concept of “target trials” as blueprints for answering causal questions. When randomized trials cannot be conducted – due to feasibility, ethics, practicality, or timeliness – trial designers can use the target trial framework to rigorously emulate the ideal randomized trial with observational or real-world data, enabling reliable answers to important causal questions.
CTTI will continue to lead efforts to modernize practice and build shared standards, helping foster truly fit-for-purpose approaches.
CTTI Presentation and Engagement Highlights
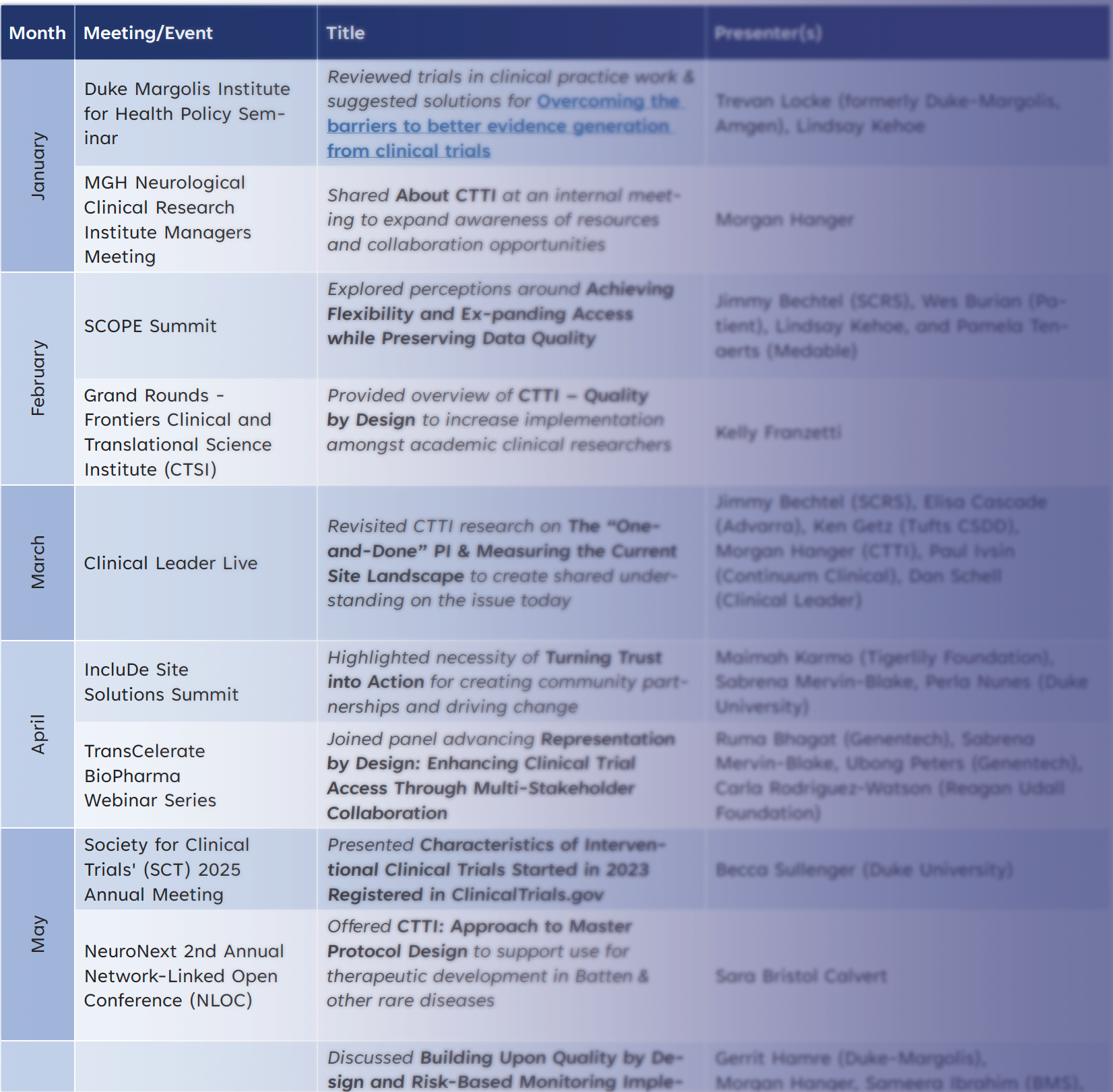
Over the past year, CTTI delivered several presentations, panels, and discussions across the clinical research ecosystem. This table below highlights where and how we shared CTTI's work and contributed to key conversations. It spans leading forums in academia, industry, patient advocacy, and policy, and advances themes like Quality by Design, accessibility, and flexible trial approaches. Thank you to our project team leaders and members, colleagues at related initiatives, and others who helped share CTTI’s resources and our commitment to improving the quality and efficiency of clinical trials.